views
Biologics are a newer class of drugs that target specific molecules involved in the immune response. The two biologics approved for atopic dermatitis are dupilumab and tralokinumab. Dupilumab is a monoclonal antibody that targets the IL-4 and IL-13 pathways, which are involved in the immune response. It was approved by the US FDA in 2017 for the treatment of moderate to severe atopic dermatitis in patients aged 12 years and older.
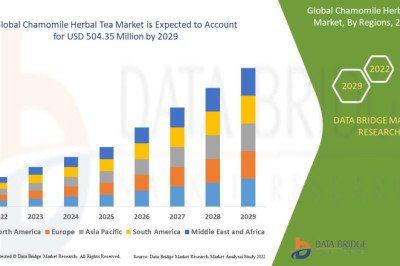
Tralokinumab is a monoclonal antibody that targets IL-13, which has been shown to be involved in the pathogenesis of atopic dermatitis. It is currently in phase 3 clinical trials for the treatment of moderate to severe atopic dermatitis. The global Atopic Dermatitis Drug Market is expected to grow at a compound annual growth rate of 4.6% from 2021 to 2028, according to a report by Grand View Research. The market was valued at $6.6 billion in 2020 and is projected to reach $9.3 billion by 2028.
Read more @ https://influentialblogging.blogspot.com/2023/02/global-atopic-dermatitis-drug-market-is.html