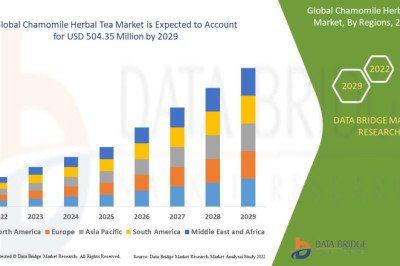
views
Studies on bioavailability and bioequivalence: The amount of a medicine that reaches the target tissue after administration is referred to as bioavailability. To ascertain whether two or more medication formulations are similar in terms of their bioavailability, bioequivalence tests are carried out. To determine a medicine's bioavailability and bioequivalence, Bioanalytical Testing Services can examine the pharmacokinetics of the drug in biological samples. PK/PD Investigations: Pharmacokinetics/Pharmacodynamics (PK/PD) studies are carried out to comprehend the connection between a drug's body-concentration and its therapeutic action. To enhance PK/PD modelling and simulation, bioanalytical testing services can quantify the pharmacokinetics and pharmacodynamics of medicines in biological samples.