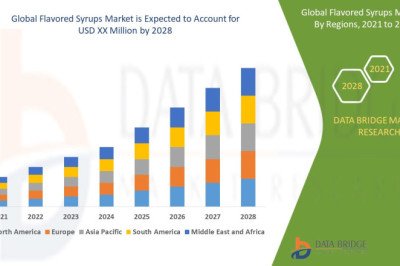
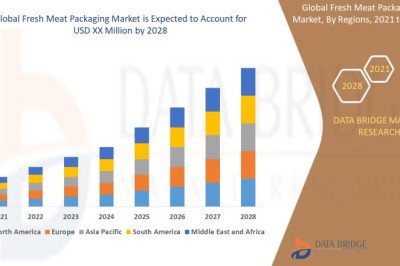
views
Lipoprotein peroxidation, particularly the modification of apolipoprotein B-100, has been implicated in the pathogenesis of atherosclerosis. However, little is known about the chemical mechanism by which apolipoproteins are derivatized during oxidation. In this study, we provide evidence that the formation of the toxic pollutant acrolein (CH2=CH–CHO) and its conjugates with lysine residues is involved in the oxidative modification of human low-density lipoprotein (LDL). After incubation with LDL, acrolein preferentially reacts with lysine residues. To determine the structure of acrolein-lysine adducts in proteins, the reaction of acrolein with lysine derivatives was performed. Using Nα-acetyllysine, we detected a single product identified as a novel acrolein-lysine adduct Nα-acetyl-Nε-(3-formyl-3,4-dehydropiperidinyl ) lysine. Acid hydrolysis of the adduct produces derivatives detectable by amino acrolein acid analysis. It was revealed that the disappearing lysine residues were partially recovered by Nε-(3-formyl-3,4-dehydropiperidinyl)lysine after in vitro incubation of LDL with acrolein. Furthermore, we found that the same derivative was detected in LDL oxidatively modified with Cu2+ and that adduct formation was consistent with LDL peroxidation assessed by consumption of α-tocopherol and cholesteryl ester and concomitant formation of cholesteryl ester hydroperoxide relevant. An enzyme-linked immunosorbent assay measuring free acrolein showed that a large amount of acrolein was released from Cu2+-oxidized LDL. Furthermore, the metal-catalyzed oxidation of arachidonic acid was associated with the formation of acrolein, suggesting that polyunsaturated fatty acids, including arachidonic acid, represent a potential source of acrolein generated during LDL peroxidation. These results suggest that acrolein is not only a pollutant but also a lipid peroxidation product that can be ubiquitously produced in biological systems.