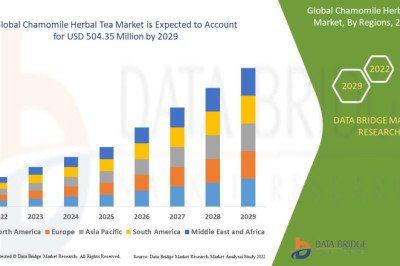
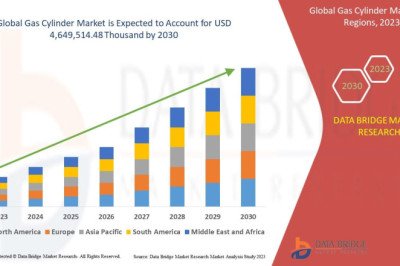
views
A natural or organic substance with the chemical formula (CH2O)2CO is ethylene Carbonate (EC). With a pleasant smell, ethylene carbonate is a translucent to yellowish solid. The interaction of ethylene oxide with carbon dioxide results in ethylene carbonate. Ethylene carbonate is often used in lithium battery electrolyte solution and as a reactant in the manufacture of polycarbonate diol because it is an exceptionally polar solvent and dissolves a significant amount of electrolyte.
To decrease the viscosity and melting point of certain electrolytes, further components can be added, such as methyl acetate, diethyl carbonate, ethyl methyl carbonate, and dimethyl Carbonate. In addition to being employed in the manufacture of polymers and other organic materials, ethylene carbonate is also used as a plasticizer and as a precursor to vinylene carbonate. In addition, it can quickly dissolve polymers that are being used as detergents and releasing agents.